What is Colloidal Silver?
A colloidal silver is a type of mix of a liquid and a solid, where tiny particles, of a particular size, are suspended in a liquid. The thing about a colloid is that the particles are very regularly sized. They aren’t as big as a suspension, but larger than those in a true solution. The trick to making the colloid is getting this sizing right. In a colloidal solution, the particles always remain dispersed evenly – they never settle to the bottom or top of the mix.
What Can You Use if for?
Silver is a well known antibiotic/antiseptic. It is used in various guises, such as would dressings. It was first used for wound dressing in 1891 by the surgeon, B.C. Crede. It was then used, up to 1940, by doctors in the USA, to treat corneal ulcers, puerperal fever, tonsillitis, and other infections.
The antibiotic effects mean that is has been used to treat various infections, such as Lyme disease and pneumonia. It is also used to treat warts and infections like ringworm.
In fact, colloidal silver has been used to treat diseases as diverse as dermatitis and appendicitis. However, note that there is little medical evidence to support many of these claims.
Used topically, it can be used to treat burns, staphylococcal infections, fungal infections, eye infections and acne.
Taken internally, it can be used for tuberculosis and other lung infections.
How Colloidal Silver Works
The silver acts on the metabolism of the bacteria, the silver binds to the cell membrane of a bacterium, preventing cell respiration and killing the bacterium.
Warnings
Taking colloidal silver internally can cause a condition called argyria. The result is a bluish tinge to the skin caused by deposits of silver compounds like silver sulfide. The compounds are formed when silver is subjected to UV light. The compounds are then deposited in tissues. Argyria doesn’t seem to have any serious health issues, other than depletion of selenium (which is a trace element needed for good immune system function).
Read more about argyria here.
The Silver Safety Council have worked out some guidelines on how much silver is safe to take and that will avoid effects like argyria.
Occasionally, you can get kidney damage from taking colloidal silver internally, so watch out for any adverse signs.
Colloidal silver can have adverse interactions with various antibiotics, such as tetracycline and certain penicillins.
How to Make Colloidal Silver
The trick to making effective colloidal silver is to minimize the number of free silver ions present in the colloid mix.
There are two basic methods to making nano-particles, for colloidal silver. Which you use depends on how much ‘chemistry’ you want to do. The first is based on electrolysis, so you’ll need some sort of battery to produce electricity; the second needs a bit of chemistry.
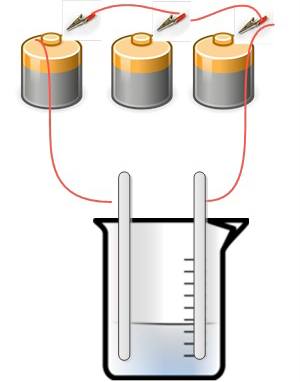 Using Electrolysis to Make Colloidal Silver
Using Electrolysis to Make Colloidal Silver
What you need:
- Silver rods for electrodes – the silver must be pure, sterling silver is not good enough
- Batteries (to generate around 27 – 30 volts) OR AC/DC electric charger rated at around 100 mA/h
- Distilled (pure) water
- Glass beaker or container – ideally borosilicate based
Method:
This is done ideally in the dark and in colder temperatures (around 39 F – 50 F). Place the silver rods into the beaker of distilled water. Connect the batteries together in a series and attach the remaining unconnected terminals to each silver rod.
It takes about 10-15 minutes before anything happens. Then you’ll see a yellowish smoky mist appear. It takes around 30 minutes for the reaction to complete.
Related: You Pass by This Plant Everyday Without Knowing How to Use It When SHTF
A Bit More Chemistry to Make Colloidal Silver
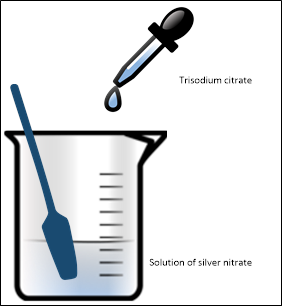 What you need:
What you need:
- Silver nitrate (1.7g dissolved in 1.75 pints of distilled water)
- Trisodium citrate (1% solution)
Method:
- Take 50ml of a silver nitrate solution and heat until boiling
- Slowly add, drop by drop, the trisodium citrate solution to the silver nitrate
- Stir vigorously
- You’ll notice a color change to yellow
- Remove from heat and stir until cooled to room temperature
To make sure that the reaction is complete take a sample from your solution and add salt (sodium chloride). If the solution turns turbid with the addition of the salt, you still have ionic silver present. If it remains clear the reaction is complete.
Related: 5 Chemistry Experiments For Preppers
Storing Colloidal Silver
You should store your colloidal silver in a dark glass bottle to avoid oxidation through sunlight.
You may also like:
 The Only 4 Antibiotics You’ll Need when SHTF
The Only 4 Antibiotics You’ll Need when SHTF
Do You Make These Fatal Mistakes In A Crisis? (video)
How to Heal Cavities After the SHTF






















I have been using Colloidal Silver for a year. I have non contractual TB and this silver has put it in remission. I will have another blood test in the near future to see how will silver demolished it!
I use it on nail fungus and it works. I have not had a cold, flu, etc since I have used the silver. How great is that! Praise God for natural medicines He provided.
Dr Mercola just had an artcle today too about silver. Go to Mercola.com
Your info is a bit off
You MUST use D.C. Power
Because batter power wears down, an electric 30 mA is best
It will take about 4 hours per liter. You have a decent solution when a red laser beam appears in the water when shown thru the solution
It could take about 24hours after the power is disconnected befor the silver ions actually appear under the laser and an ideal solution is a pale yellow
My first attempt to make colloidal silver was with 2 1 oz .999 silver bars from Silver Towne. I make my own distilled water and know it it pure. I used 3 9 v batteries and suspended the bars 1.5 inches from each other. After about an hour the water was cloudy and very grey. One of the bars was absolutely black and there was a black sediment on the bottom of the jar. I am very discouraged. Any suggestions?
You cooked it to long put very light salt solution in distilled water 1/4 teaspoon salt to 8 oz water disalved then use 2 drops for 16 oz will only take about 20 min to cook water will look a little cloudy.
Don’t leave it so long. 30 minutes is sufficient.
DO NOT USE SALT
Can we produce colloidal gold by the same way ??
I have been making colloidal silver for YEARS. Do NOT add salt. The easiest way is just to add a few ozs of colloidal from your last batch to the distilled water. This lets the process start immediately. The silver turning black is normal. No problem. When finished I just pour the batch through a paper coffee filter to get the particles out of the batch, although the do no harm.
Also silver towne has been copied. ie a reproduction bar would not be silver. Perhaps get scan on silver.
yeah sir and drink
tunerfish —
I guess the only thing you did correctly is try. I’ll try to get you on track. I’ve been making colloidal silver for 20 years.
First off — you only need 2 pure silver wires about 12 gauge and 6 inches long.
Second — Electric current doesn’t pass through distilled water very well. You need to dissolve a pinch of sea salt in about 2 ounces of distilled water. Using a dropper, put 4 drops of the salt water in 16 ounces of distilled water and stir. Then place your wires that are attached to your batteries in the water.
Third — DO NOT leave this running for an hour. With fresh batteries you will get about 12 to15 ppm (parts per million). That is a standard mix.
Fourth — For additional information contact The American Sentinel, 704-504-1899. They also sell a regular colloidal silver generator. If you really want the best — they now have a Nano-Particle silver generator. It’s not cheap but the particles can actually penetrate a cell and kill all the bad stuff.
Good luck. Mike
tunerfish —
ooops — it actually only needs to run about 15 minutes to get the correct ppm.
Good luck, Mike
Thanks for all your efforts that you have put in this.
Very interesting info.
will a silver Mexican coin work ,where do I get the correct type of silver from, the cheapest amount ,to do this ,and isbottled water ok to use
I want to make colloidal silver. the type that will be able to penetrate the cells in your body is this doable, and how do I do it , and what do I need to do this that is affordable thanks tom
I hav been using silver for bout a yr. Now I’m amazed at it’s uses. I began using it after a trip to the hospital for diverticulitis. OMG the pain. Anyway the antibiotics they gave me caused horrible side effects of permanent damage to muscles tendons an cartilge . After 5days in hospital I came home with mersa. Even though I was. On antibiotics for the diverticulitis the mersa continued. Back to dr.s an more antibiotics an could not stop the mersa. I had 6boils under each armpit OMG. Long story short I began soaking in Epsom salts hot hot water this is after I sprayed sores with novicane I then took silver internally and washed externally with it in couple days u could see it was working. I did this 3x a day in beginning. The sore I had on my leg was eating away flesh turned blk. I thought they’d cut my leg off. But I persisted. It took couple weeks but thre silver healed it.
Since then I have used silver in water to kill a fungus on my baby tree. I’ve used it to heal ear I fection in the dog.ive used it when virus was spreading through work place. An today I’m going to try it on the bad spot in the lawn.
I also found another product that heals the diverticulitis called oreganol, the liquid form,. As with anything be careful use wisdom, faith too, and legally have to say ask your Dr.
I’ve used it for over 20 years .yes you can make it like they show but Theresa better way get a 15/30/ volt DC transformer about on e amp .use only the 30 volt switch .heat your water about a quart to boiling then place your electrodes in it .I use hook or bent silver .999 wire you can by on net only use .999 or better .then dry off the table put a white paper towel under pot of water and use a coffee pot glass it’s borosilicate glass doesn’t break .now drop about ten grains of salt between the electrodes that are about half inch apart .the hot water accelerates the production watch the pot with a light for a yellow color to just start forming then shut it down .it will continue to turn yellow while cooling down .clean your electrodes with a stainless steel micro brush .it’s quick this way like 20 minutes .store in dark .put in spray bottle and spray a stinky empty garbage can .spoke it well then smell ..no smell .just like rain smell .I use it for rain water I drink one ounce per five gal.wait one hour then use.it’s really less time but why chance it I’ve never gotten sick from water with it . jim PS if you use hot water with the 9 volts battery set up it will destroy them .like it’s a dead short .transformer last a lifetime and more .
Can anyone tell me how to make colloidal selenium .?it kills cancer big time .
My family and I have been using colloidal silver for 8 years. We have not needed any other medication or pharmaceutical from Big Pharma.
Don’t forget, that most of the men in the first World War were treated with colloidal silver and silver powder.
In the 1960’s, new born babies had silver powder sprinkled on their belly buttons to prevent infection. My wife still has her little bottle of powder.
The main reason for words being said against it is that Big Pharma can not patent it, or charge for it.
Our usage of colloidal silver amounts to around a few pennies each year… as opposed to £1500 of chemical costs via Doctors.
As a final couple of comments… Alexander the Great had his water carried in silver vases, and that was a really long time ago. As for being born with a silver spoon in your mouth? Eating your food with a silver fork and spoon meant that you would absorb some fraction of them in to yourself when you ate, and they would not hold any infection while stored!!!
Silver has been used for time immemorial.
Is 10 gage rods good for making 30ppm, and is time longer due to the larger gage?
I am wondering if using a current limiting voltage source would be beneficial in order to keep the particle size consistent, and if so, what would be the best current setting?
Is it possible to use a 9 volt dc.charger
Instead of batteries, I’m a poor man and that would be cheaper for me
Happens to all of us when we start making CS but no problem.
Let it settle a bit take out the rods and filter the solution through a coffee filter. The rods can be wiped down from time to time to clean them OR if you have an air pump just put it in when you start the process and keeping it agitated keeps the rods clean and seems to make a better product.
Is there anywhere you can just buy this stuff without having to make it and how do you know how much is safe to injest?
You can buy it at Health Foods stores or online. I started making it myself because it’s so cheap to make at home. The stores want about $85 a quart. I use an A/C Adapter with an output of 1.3 VDC – 1.3A. Cut off the original charge plug and strip an inch off each of the 2 wires coming out of the Adapter – attach each to an alligator clip. I use a 2 quart glass vase – fill with distilled water to 1/2 inch from top. Put a plastic cover from a coffee can on top(cleaned) make 2 slits in plastic – then use 2 -1 ounce pure silver bars put into each slit with half of the silver bars in the water. Attach the alligator clips to the top of silver bars – plug into outlet. I use a small fish tank aerator to circulate the water. I have been doing this Safely for 8 years now. It takes about 12 hours for that batch. I rotate the silver bars every 2-3 hours – especially when the bars turn black.
It is not dangerous to injest. However, it is not effective under 100 ppm. I am not sure how it is done but there is a method that will transfer the silver proteins only.. it is the silver salt that when taken internally can cause argyria which is not dangerous, just weird. Silvadine cream (widely used for burns) is a silver salt compound. When used externally also will not cause argyria.
Actually Stephanie, the industry agreed upon is the sweet spot between 10-20PPM.
Any more than that, the particles will get larger and larger making it far less effective.
Science has proven that the smaller the particles the more effective.
My new machine makes CS with particles down to .00008 nanometers!
I encourage you to keep reading. The silveredge.com has a lot of good info even though I didn’t buy mine there. (A scientist told me they have good info.)
I don’t sell this machine but being overbuilt to last and only 158.00 I can’t be more enthusiastic about it. I add an air stone to the air tube putting oxygen into the water and can do a whole gallon in 5 hours. That is between 10-11.5 PPM.
OK: ADD SALT DO NOT ADD SALT….
I’M CONFUSED!
DO NOT add SALT!
I’ve been using colloidal silver for years now, though, admittedly I purchase it instead of trying to make my own (though I do know how to make it, I just don’t have access to pure silver at the time). It’s an antimicrobial, not just an antibiotic. This means it works on bacteria, viruses and fungi that occur in the human body (and apparently trees, too, per a response above)!
When the rona outbreak started, I actually had some pre-mixed with a capful of hydrogen peroxide in a standard size saline nasal spray bottle (that I emptied and thoroughly cleaned) to snort anytime I had to go out in public. Works like a charm to help prevent nasties caused by inhalation through the nasal passages as well as chronic sinus issues from allergies!
I’ve used CS on surgical and accidental wounds and they’ve healed with no infection or issues. I use it on one of my cats who gets a chronic acne by her tail and it keeps her clear of outbreaks and her fur is finally growing back… I add a capful to distilled water that I give to my kitties, keeps the sneezies and sniffles away, and I’d think it would work on various parasites like ring and round worms if they went outside and were exposed to such things.
I think everyone should have a quart or two of this on hand for everyday use, not just for when SHTF. It’s an amazing supplement to use to help keep many illnesses and infections away. Just try to get the highest quality CS you can find. I usually get stuff that’s 20k PPM.
DO NOT use salt. Just have a little patience.
All right, I have to know. But I don’t need to know so badly that I run an experiment with boiling hot water, electricity and a handful of salt. So, knowing enough Chemistry I know that Silver can be quite…difficult (which I first found out about in 8th grade Science where we had been using Silver Nitrate soln and the teacher said that if you got ANY on your skin to wash it off very well. I found out that soap and water doesn’t always guarantee that whatever you wanted removed has been done so. Nothing happened for another 10 minutes…until I got out into direct sunlight: my fingers turned an unnatural coal-black and very dark grey that took 2 days to remove! I also have seen what happens when you get real sodium that cuts like cold butter and burns your skin. I dropped my piece that my friend had cut off and handed me (who was wearing gloves) and so I went to retrieve my metal in the snow but my friend yelled, “NO!” and pulled me back just in time to see and hear a loud pop. Sodium and Chlorine are a perfect pair as Sodium has an electron it really does not want, but Chlorine wants more than anything just one more electron! So I would hate to think what might happen if sodium is willing to trade with anything other than their perfect mate. But I still have to know. Please. I would surmise that because of the 2 different people who said to dissolve a small amount of salt and water (distilled, always!) and add a few drops of the weak saline soln to the hot water and silver bath that may or may not still have current running through it.
The other thing is availability of resources. Namely, .999 Ag wire. So, am I correct in assuming that silver wire as found in a craft store is not pure (. 999) Silver? I know Sterling is not even close to pure! Something like 92% Silver? And American Silver dollars, the good ones that are $90 USD and up: are they .999 or at least .990? Thank you in advance!
Has anyone ever benefited from taking regular doses of CS for tinnitus or ringing of the ears?